Quiz-summary
0 of 1 questions completed
Questions:
- 1
Information
Question description.
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading...
You must sign in or sign up to start the quiz.
You have to finish following quiz, to start this quiz:
Results
0 of 1 questions answered correctly
Time has elapsed
Categories
- Not categorized 0%
- 1
- Answered
- Review
- Question 1 of 1
1. Question
N2(g) + 3H2(g) ⇌ 2NH3(g) ; ΔH° = -92.22 kJ•mol-1
Which change would decrease the Keq in the above reaction?
CorrectThe trick to this question is to realize the only thing that can change the Keq is changing the temperature, thus Choice [E] must be the correct answer. All the other answers will not affect the equilibrium or only affect the reaction quotient, Q.
The easiest way to think about the ΔHo of a reaction is to add it into the chemical equation. A negative ΔHo means the reaction is exothermic, so energy is given off, or it is part of the products. The reverse is true for endothermic reactions, the energy would be part of the reactants. The chemical reaction can be rewritten as:
N2(g) + 3H2(g) ⇌ 2NH3(g) + energy
Next, we have to understand the concept of Keq. You can think of Keq as the concentration of products divided by the concentration of reactants, or:
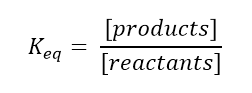
Decreasing the Keq means we are increasing the amount of reactants, so we have to find which answer will shift the equilibrium towards the reactants, or towards the left.
Increasing the temperature will shift the equilibrium towards the left, or towards the reactants, and thus decrease the Keq.
Adding a catalyst does not change the Keq. Decreasing the volume of the vessel will increase the pressure, and the reaction will shift towards the side that has less moles of gas to reach equilibrium, or in this case towards the products, thus increasing Q. Injecting H2 gas will push the equilibrium towards the products, increasing Q. Lastly, cooling ammonia gas into liquid will pull the reaction towards the right to produce more products, increasing Q.
Extended Explanation – useful if you need more review
IncorrectThe trick to this question is to realize the only thing that can change the Keq is changing the temperature, thus Choice [E] must be the correct answer. All the other answers will not affect the equilibrium or only affect the reaction quotient, Q.
The easiest way to think about the ΔHo of a reaction is to add it into the chemical equation. A negative ΔHo means the reaction is exothermic, so energy is given off, or it is part of the products. The reverse is true for endothermic reactions, the energy would be part of the reactants. The chemical reaction can be rewritten as:
N2(g) + 3H2(g) ⇌ 2NH3(g) + energy
Next, we have to understand the concept of Keq. You can think of Keq as the concentration of products divided by the concentration of reactants, or:
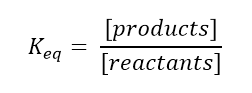
Decreasing the Keq means we are increasing the amount of reactants, so we have to find which answer will shift the equilibrium towards the reactants, or towards the left.
Increasing the temperature will shift the equilibrium towards the left, or towards the reactants, and thus decrease the Keq.
Adding a catalyst does not change the Keq. Decreasing the volume of the vessel will increase the pressure, and the reaction will shift towards the side that has less moles of gas to reach equilibrium, or in this case towards the products, thus increasing Q. Injecting H2 gas will push the equilibrium towards the products, increasing Q. Lastly, cooling ammonia gas into liquid will pull the reaction towards the right to produce more products, increasing Q.
Extended Explanation – useful if you need more review
