Quiz-summary
0 of 1 questions completed
Questions:
- 1
Information
Question description.
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading...
You must sign in or sign up to start the quiz.
You have to finish following quiz, to start this quiz:
Results
0 of 1 questions answered correctly
Time has elapsed
Categories
- Organic Chemistry 0%
- 1
- Answered
- Review
- Question 1 of 1
1. Question
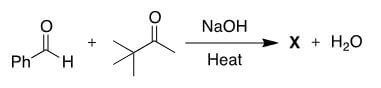
What is the structure of the major product (X) of the following aldol cross-condensation?
 Correct
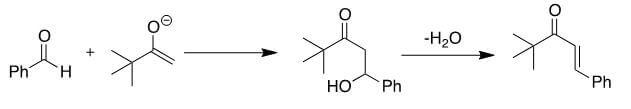
CorrectAldol condensations involve the formation of an enolate, followed by an attack on a carbonyl carbon. In this case, there is only a single α-carbon that has an acidic hydrogen. This means that only one of the two reactants can form an enolate, as shown below. The enolate will attack the aldehyde and form a β-hydroxyketone, shown in the center below. This molecule will undergo a dehydration to form the more thermodynamically stable trans-α, β-unsaturated ketone [B].
 Incorrect
IncorrectAldol condensations involve the formation of an enolate, followed by an attack on a carbonyl carbon. In this case, there is only a single α-carbon that has an acidic hydrogen. This means that only one of the two reactants can form an enolate, as shown below. The enolate will attack the aldehyde and form a β-hydroxyketone, shown in the center below. This molecule will undergo a dehydration to form the more thermodynamically stable trans-α, β-unsaturated ketone [B].

